The reason we haven't heard about it is that this condition acts imperceptibly, usually doesn't raise too many alarming signals, and until recently, doctors didn't even consider it on their radar. But we'd be surprised if we listed all the diseases and conditions caused by chronic inflammation. For these reasons, in recent years, numerous scientists have dedicated themselves to studying this condition and its interrelationships with major chronic diseases such as diabetes, cancer, depression, Alzheimer's, and obesity, to name a few.
But what do we mean specifically when we talk about chronic inflammation?
In Latin, the word inflammatio means "to ignite," "to set on fire." Our body "inflames," "ignites," and "catches fire" as a response to an attack. The most common attacks are infections and bacteria. Our body's reaction to these types of attacks is fairly well-known, as most of us have suffered an infection or insect bite at some point. Whatever the case, we've surely experienced some of the characteristic symptoms of inflammation, such as sharp pain, swelling, redness, a sensation of "heat," and even loss of function in the affected area.
This type of inflammation acts locally, appearing quickly in response to an attack, and subsides when our body has managed to eradicate the threat and repair the damage.
Since the beginning of time, our bodies have been exposed to environmental aggression. Since we didn't have access to antibiotics or modern medicine for most of our evolution, we had to develop our own defense mechanisms. For this reason, our bodies are already equipped with self-regulating biological processes that act automatically whenever we need to repair a wound, cure an infection, or fight off bacteria. In these cases, specific cells of the immune system are activated when they detect that our body is being attacked by an external agent (i.e., a virus or bacteria). Based on this signal, our body coordinates a synchronized response that also involves the circulatory and vascular systems, our endocrine system, and various mediators such as white blood cells (leukocytes), mast cells (which release a chemical called histamine that helps repel the action of various pathogens), and macrophages (which release cytokines, a type of pro-inflammatory protein that reinforces the immune system's defensive action).
So far there are no major problems.
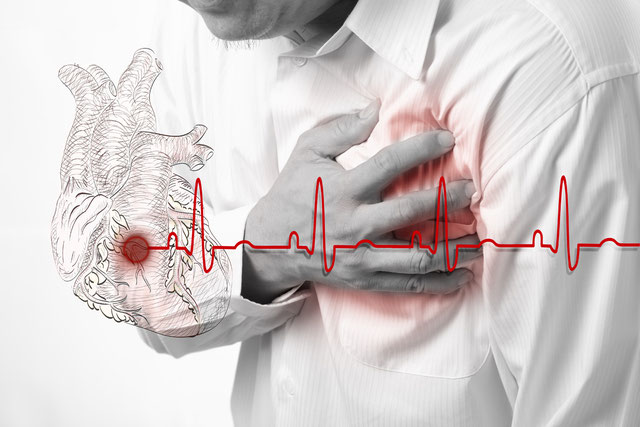
But there is another type of inflammation that isn't visible, where the characteristics of acute inflammation (i.e., redness, swelling) aren't observed, and where our immune system's response doesn't subside once the threat has disappeared. When inflammation ceases to be localized and becomes systemic, our body remains "on" even when it's not under attack. Under normal conditions, the activation of the immune response and the release of various mediators such as cytokines is beneficial, but when this response remains active and cytokine production becomes chronic, it ends up causing other problems. The danger lies in the fact that chronic inflammation continues to do its work and doesn't become visible until the person suffers an episode. An example is people who suffer a sudden heart attack despite not having felt any discomfort or warning signs beforehand.
The first to observe the effects of systemic inflammation were cardiologists, who identified the presence of markers associated with chronic inflammation in people with cardiovascular disease.

But why should we care?
It turns out that chronic inflammation is a source of oxidative stress. We've already explained how oxidation and the action of free radicals damage our cells (and mitochondria), accelerate the aging process, and also how they affect our brain's homeostasis. And not only that. As we mentioned at the beginning of this section, chronic inflammation is capturing the attention of many researchers as it is present in a range of very common diseases such as cancer, depression, diabetes, and Alzheimer's, to name a few.
Take the relationship between chronic inflammation and obesity as an example.
In obese people, an "abnormal" increase in systemic cytokine concentrations is often observed, especially those of the types TNF-α (tumor necrosis factor-alpha), IL-6 (interleukin-6), and CRP (C-reactive protein). In these cases, overactivation of the immune system occurs in response to fat accumulation, especially adiposity in the abdominal area. In obese people, excess fat ends up being deposited in areas not designed for that purpose. Immune cells mistake fat deposits for intruders or aggressors that must be attacked. The excessive release of cytokines ultimately alters the balance of other chemicals and some hormones that regulate appetite, such as leptin, which regulates the feeling of satiety. This is the beginning of a vicious cycle, since when inflammation becomes permanent, obese people end up developing leptin resistance. From that moment on, the body cannot feel satiated even shortly after eating. It is known that obese people are more prone to cardiovascular attacks. As expected, overweight people tend to have markers associated with inflammation at levels higher than "normal," and the amount of cytokines in their body is often double or triple the levels observed in non-overweight people.
One of the markers used to measure the degree of systemic inflammation is the level of the C-reactive protein (CRP), which is produced in the liver as part of the immune response. This blood test uses a variation expressed as hs-CRP (high-sensitive C-reactive protein). Low levels of hs-CRP are essential for preventing cardiovascular events and the development of Alzheimer's disease. High levels (>3.0) of hs-CRP triple our risk of coronary and cardiovascular disease (i.e., arteriosclerosis), independently of other risk factors.
Let's take another example, in this case the relationship between chronic inflammation and depression. For some researchers, it's no coincidence that the characteristic symptoms of depression, such as lack of motivation, fatigue, and mental lethargy, are the same symptoms a person experiences when they are "sick." If sick people and depressed people "feel" and "behave" similarly, there should be some common denominator between them. Apparently, that common denominator would be chronic inflammation and the excessive release of cytokines when they are not needed. Various scientific studies have attempted to validate this relationship and found that there are higher amounts of cytokines during depressive episodes, and vice versa during remissions. In other similar studies, where bacteria were injected into healthy people to induce depressive states, it was observed that the immune response and cytokine release coincided with the activation of brain areas associated with depression. These findings are reinforced by cases observed in people with rheumatoid arthritis and multiple sclerosis, where depression is very common. Depression is also observed as a fairly common side effect in cancer patients receiving alpha interferon. There is speculation that depression arises due to the pro-inflammatory effects of interferon.
Chronic inflammation is also an underlying characteristic in people with high stress levels and the reason why prolonged stress inevitably leads to depression. Following these findings, many practitioners have introduced the use of anti-inflammatory herbs and nutrients such as curcumin and omega-3 essential fatty acids (EPA/DHA) as part of their pharmacological arsenal for treating depression.
The best way to control chronic inflammation and avoid its negative effects is to eliminate its underlying causes. These causes are deeply rooted in our habits and lifestyle, our diet, and our exposure to environmental toxins, so there are many things we can do.

What does inflammation in the body have to do with the risk of developing Alzheimer's disease, suffering a heart attack, or succumbing to colon cancer?
More than you might think. As scientists delve into the root causes of these and other diseases, they are beginning to see links to an immune defense mechanism called inflammation—the same biological process that turns the tissue around a wound red and causes swelling in an injured toe. If they're right—and the evidence is starting to become very strong—it could radically change doctors' understanding of what makes us sick. It could also be a boon for pharmaceutical companies looking for new ways to keep us healthy.
More often than not, inflammation is a lifeline that allows our bodies to defend themselves against various diseases caused by bacteria, viruses, and parasites. (Yes, even in the modern world, we are constantly bombarded by pathogens.) The moment any of these potentially deadly microbes enter the body, inflammation mounts a defensive attack that devastates the invaders and any tissue that may have been affected. Then, just as quickly, the process subsides, and healing begins.
Occasionally, however, all that fervent production doesn't shut down at just the right time. Sometimes the problem is a genetic predisposition; other times, actions like smoking or high blood pressure keep the process going. In either case, the inflammation becomes chronic rather than transient. When that happens, the body turns on itself—like a stubborn child who can't resist touching a wound—with consequences that seem to underlie a wide variety of diseases.
Suddenly, inflammation has become one of the hottest areas of medical research. Not a week goes by without the publication of another study uncovering a new form of chronic inflammation that damages the body. It destabilizes cholesterol deposits in the coronary arteries, leading to heart attacks and even potentially strokes.
Nerve cells are lost in the brains of Alzheimer's victims. This can even promote the proliferation of abnormal cells and facilitate their transformation into cancer.
In other words, chronic inflammation may be the driving force behind many of the most feared diseases of middle and old age.
This concept is very exciting because it suggests a new and possibly much simpler way to protect against disease. Instead of different treatments for, say, heart disease, Alzheimer's, and colon cancer, reducing inflammation could be a single remedy that prevents all three. Chronic inflammation also fascinates scientists because it indicates that our bodies can, from an evolutionary perspective, become victims of their own success. "We evolved as a species because of our ability to fight off microbial invaders," says Dr. Peter Libby, chief of cardiovascular medicine at Brigham and Women's Hospital in Boston. "The strategies our bodies use for survival were important at a time when we didn't have plants to purify water, and when we didn't have sewage systems to protect us."
But now that we're living longer, those inflammatory strategies are more likely to slip out of our control. To make matters worse, it appears that many of the attributes of a Western lifestyle—such as a diet high in sugars and saturated fats, accompanied by little to no exercise—also make it easier for the body to become inflamed.
The new view of inflammation is changing the way some scientists conduct medical research. "Virtually all of our R&D efforts are focused on inflammation and cancer," says Dr. Robert Tepper, president of research and development at Millennium Pharmaceuticals in Cambridge, Massachusetts.
In medical schools across the U.S., cardiologists, rheumatologists, oncologists, allergists, and neurologists are talking to each other—and discovering they're looking at the same thing. The speed with which researchers are jumping on the inflammation bandwagon is breathtaking. Just a few years ago, "no one was interested in this stuff," says Dr. Paul Ridker, a cardiologist at Brigham and Women's Hospital who has done some of the pioneering work in the area. "Now the whole field of inflammation research is about to explode."
Meanwhile, another group of sentinels, called macrophages, begins an immediate counterattack and releases more chemicals, called cytokines, signaling for more reinforcements. Soon, wave upon wave of immune cells floods the site, destroying pathogens and damaged tissue alike—there's no retreating the wounded from the battlefield in this war. (No wonder the ancient Romans compared inflammation to being under fire.) Doctors call this generalized response to virtually any kind of attack innate immunity. Even the bodies of animals as primitive as starfish defend themselves this way.
But higher organisms have also developed a more precision-guided defense system that helps direct and intensify the innate response and creates specialized antibodies, tailor-made to target specific types of bacteria or viruses. This so-called evolutionary immunity is what allows pharmaceutical companies to develop vaccines against diseases like smallpox and influenza. Working together, the innate and evolutionary immune defenses fight pitched battles until all invading germs are annihilated. In a final burst of activity, a final wave of cytokines is released, the inflammatory process recedes, and healing begins.
Problems begin when, for one reason or another, the inflammatory process persists and becomes chronic. The final effects are varied and depend greatly on which part of the body the uncontrollable reaction takes hold.
Among the first to recognize the broader implications were heart doctors who realized that inflammation appears to play a key role in cardiovascular disease.
Is your heart at risk?
Not long ago, most doctors thought of heart attacks primarily as a plumbing problem.
Over the years, fatty deposits gradually accumulate inside the main coronary arteries until they become so large that they cut off the blood supply to a vital part of the heart. A complex molecule called LDL, the "bad" cholesterol, is always the raw material for these deposits.
It was clear that anyone with high LDL levels was at greater risk of developing heart disease.
There's just one problem with this explanation: sometimes it's completely wrong. In fact, half of all heart attacks occur in people with normal cholesterol levels. Not only that, but with improved imaging techniques, doctors discovered, to their great surprise, that the most dangerous platelets weren't necessarily large. Something that hadn't yet been identified was causing the deposits to burst, producing large clots that cut off coronary blood supply.
By the 1990s, Ridker became convinced that some sort of inflammatory reaction was responsible for the platelet breakdown, and he set about trying to prove it. To test his hunch, Ridker needed a simple blood test that could serve as a marker of chronic inflammation. He settled on C-reactive protein (CRP), a molecule produced by the liver in response to an inflammatory signal. During an acute illness, such as a severe bacterial infection, CRP levels quickly skyrocket from less than 10 mg/L to 1,000 mg/L or more. But Ridker was more interested in the low levels of CRP—less than 10 mg/L—found in healthy people and which indicate only a slightly elevated level of inflammation. In fact, the difference between normal and elevated is so small that it must be measured using a specially designed, highly sensitive CRP test.
In 1997, Ridker and colleagues at Brigham and Women's demonstrated that healthy middle-aged men with the highest CRP levels were three times more likely to suffer a heart attack in the next six years than those with the lowest CRP levels. They eventually determined that inflammation resulting in a CRP reading of 3.0 mg/L or higher can triple the risk of heart disease. The risk appears to be even greater in women than in men.
In contrast, people with very low levels of C-reactive protein, less than 0.5 mg/L, rarely have heart attacks.
Doctors still don't know for sure how inflammation can cause platelet breakdown. But they have a theory. As the level of LDL cholesterol in the blood increases, they speculate, some leaks into the walls of the coronary arteries and stays there. Macrophages, alerted to the presence of something that doesn't belong there, move in and try to clear the cholesterol. If, for whatever reason, the cytokine signals gradually increase, the inflammatory process will begin instead of decreasing, and the platelet will become unstable.
"It's not about replacing cholesterol as a risk factor," Ridker says. "Cholesterol deposits, high blood pressure, smoking—they all contribute to the development of underlying plaques. What inflammation does is contribute to the propensity for plaques to rupture and cause a heart attack. If there's just inflammation, but no underlying heart disease, then there's no problem." At this point, cardiologists still aren't ready to recommend the general population be screened for inflammation levels. But there's a growing consensus that CRP should be measured in those at moderately elevated risk of developing cardiovascular disease. At the very least, a high CRP level might tip the balance in favor of more aggressive therapy with treatments—such as aspirin and statins—that are already known to work.
A new vision of Diabetes
Before Dr. Frederick Banting and his colleagues at the University of Toronto isolated insulin in the 1920s, doctors treated diabetes with high doses of salicylates, a group of aspirin-like compounds. (They were desperate and also tried morphine and heroin.) Salicylate effectively lowers blood sugar levels, but at a high price: side effects include a constant ringing in the ears, headaches, and dizziness. Today, diabetes treatments are much safer and generally work by replacing insulin, increasing its production, or helping the body use the hormone more efficiently. But researchers in recent years have revisited the salicylate approach for new clues about how diabetes develops.
What we've discovered is a complex interplay between inflammation, insulin, and fat—whether in the diet or in the deep folds beneath the skin. (In fact, fat cells behave much like immune cells, pumping out inflammatory cytokines, especially as we gain weight.) How inflammation fits into this scenario—whether as a cause or an effect—is still unclear. But the case for its central role is growing stronger. Dr. Steve Shoelson, a senior investigator at the Joslin Diabetes Center in Boston, has generated a strain of mice whose fat cells are inflammation factories. The mice become less efficient at using insulin and go on to develop diabetes. "We can reproduce the entire syndrome just by inciting inflammation," Shoelson says.
This suggests that early intervention in the inflammatory process can reverse some of the effects of diabetes. Some medications already used to treat the disorder, such as metformin, may work because they also impair the inflammatory response. Furthermore, preliminary research suggests that high CRP levels may indicate an increased risk of diabetes. But it's too early to say whether lowering CRP levels can actually keep diabetes at bay.
Cancer: The wound that never heals
Back in 1860, renowned pathologist Rudolf Virchow speculated that cancerous tumors arise at the site of chronic inflammation. A century later, oncologists paid more attention to the role of different genetic mutations in promoting abnormal growths that eventually become malignant. Now, researchers are exploring the possibility that mutation and inflammation mutually reinforce processes that, if left unchecked, can transform normal cells into life-threatening tumors.
How can this happen?
One of the most potent weapons produced by macrophages and other inflammatory cells are so-called oxygen free radicals.
These highly reactive molecules destroy almost anything in their path—particularly DNA. A glancing blow that damages, but doesn't destroy, a cell could result in a genetic mutation that allows it to continue growing and dividing.
The abnormal growth isn't yet a tumor, says Lisa Coussens, a cancer biologist at the Comprehensive Cancer Center at the University of California, San Francisco. But to the immune system, it's a lot like a wound that needs tending. "When immune cells are called upon, they bring growth factors and a lot of proteins that will call upon other inflammatory cells," Coussens explains. "Those things come and go to heal, to cure, to heal." But instead of curing, what they do is 'feed, augment, provide.'"
Sometimes the reason for the initial inflammatory cycle is obvious—as with chronic heartburn, which continually bathes the esophageal mucosa with stomach acid, predisposing a person to esophageal cancer. Other times, it's less clear. Scientists are exploring the role of an enzyme called cyclooxygenase 2 (COX-2) in the development of colon cancer. COX-2 is another protein produced by the body during inflammation.
In recent years, researchers have shown that people who take daily doses of aspirin—which is known to block COX-2—are less likely to develop precancerous growths called polyps. The problem with aspirin, however, is that it can also cause internal bleeding. Then, in 2000, researchers showed that Celebrex, another COX-2 inhibitor that is less likely than aspirin to cause bleeding, also reduces the number of polyps in the large intestine. So, should you take Celebrex to prevent colon cancer? It's still too early to say. Clearly, COX-2 is one factor in colon cancer. "But I don't think it's the only answer," says Ray DuBois, director of cancer prevention at the Vanderbilt-Ingram Cancer Center in Nashville, Tennessee. "There are a lot of other components that need to be explored."

Aspirin for Alzheimer's disease?
When doctors who treat Alzheimer's patients took a closer look at those who appeared to be succumbing to the disease, they discovered a tantalizing clue: those already taking anti-inflammatory drugs for arthritis or heart disease tended to develop the disorder later than those not taking anti-inflammatory drugs.
Perhaps the immune system mistakenly saw the characteristic plaques and neurofibrillary tangles that accumulate in the brains of Alzheimer's patients as damaged tissue that needed to be cleared. If so, the ensuing inflammatory reaction was doing more harm than good. Blocking it with anti-inflammatory drugs may limit, or at least delay, any damage to cognitive function.
The most likely culprits this time are glial cells, whose function is to nourish and communicate with neurons.
Researchers have discovered that glial cells can also act much like mast cells in the skin, producing inflammatory cytokines that call other immune cells into action. "Glial cells are trying to return the brain to a normal state," explains Linda Van Eldik, a neurobiologist at Northwestern University Feinberg School of Medicine in Chicago. "But for some reason, in neurodegenerative diseases like Alzheimer's, the process seems to get out of control. You're dealing with chronic activation of glial cells, which results in an inflammatory state."
It seems that some people are more sensitive to plaques and tangles than others. Perhaps they have a genetic predisposition. Or perhaps a long-standing bacterial infection, such as gingivitis or gum disease, keeps internal activity high and tips the balance toward chronic inflammation.
Preliminary research suggests that low-dose aspirin and fish oil capsules—two of which are known to reduce inflammatory cytokines—appear to reduce a person's risk of Alzheimer's disease. Unfortunately, most of these preventative measures must be started long before neurological problems develop. "What we've learned with dementia is that it's very hard to improve those who already have it," says Dr. Ernst Schaefer, professor of medicine and nutrition at Tufts Friedman School of Nutrition in Boston. "But at least it will be possible to stabilize people and prevent the disease."
When our body attacks itself.
Doctors with the most experience treating chronic inflammation are those who specialize in rheumatoid arthritis, multiple sclerosis, lupus, and other autoimmune diseases.
For decades, these diseases have provided the clearest example of a body at war with itself. But the spark that fuels its internal destruction doesn't come from excess cholesterol deposits or a persistent bacterial infection. Instead, in a strange twist of fate, the super-sophisticated body misguides its immune defenses and directs an inflammatory attack against healthy cells in places like the joints, nerves, and connective tissue.
In recent years, powerful drugs like Remicade and Enbrel, which specifically target inflammatory cytokines, have worked wonders against rheumatoid arthritis and other autoimmune disorders. But as is often the case in medicine, the drugs have also created some problems. Patients taking Remicade, for example, are slightly more likely to develop tuberculosis, given that the same inflammatory cytokines that attacked their joints also protect them against tuberculosis.
Inflammation can be more of a problem in the early stages of autoimmune diseases like multiple sclerosis. So much tissue is eventually destroyed that the nerve damage becomes permanent. "The initial goal is to keep the immune response under control, but then you have to ask how the damaged tissue regenerates," says Dr. Stephen Reingold, vice president of research programs at the National Multiple Sclerosis Society. A solution could take decades.
Asthma without allergies?
One of the most intriguing questions in immunology today is why not everyone suffers from asthma. After all, the air we breathe is full of germs, viruses, and other irritants.
Since half of the 17 million Americans with asthma are hypersensitive to common substances like cat dander or pollen, it stands to reason that allergic reactions trigger chronic inflammation in their bodies. However, people who develop asthma as adults—one of the fastest-growing segments of the population—often don't have allergies. Doctors don't yet know what's driving their disease, but signs of inflammation are present in their lungs.
Many asthma treatments are designed to control inflammation, although they don't yet cure the disease. "It may mean that the inflammatory hypothesis isn't entirely correct or the drugs we use to treat inflammation aren't fully potent," says Dr. Stephen Wasserman, an allergist at the University of California, San Diego. "There are a lot of gaps yet to be filled." Everywhere, however, doctors are finding evidence that inflammation plays a bigger role in chronic disease than they thought. But that doesn't necessarily mean they know what to do about it. "We're in a quandary right now," says Dr. Gailen Marshall, an immunologist at the University of Texas Medical School at Houston.
"We're making progress on the idea of raising awareness. But I can't really recommend specific treatments yet." That may change soon. Researchers are looking beyond aspirin and other multipurpose drugs and toward experimental medications that block inflammation more precisely. Any day now, Genentech is awaiting an FDA decision on its colon cancer drug, Avastin, which targets one of the growth factors released by the body as inflammation gives way to healing.
Millennium Pharmaceuticals is testing a different type of drug, called Velcade, which has already been approved for the treatment of multiple myeloma, lung cancer, and other malignancies. But there is a sense that much more basic research into the nature of inflammation needs to be done before scientists better understand how to limit the damage from chronic diseases.
In the meantime, there are things we can all do to curb our inflammatory fires. Some of the advice may sound awfully familiar, but we have new reasons to keep going. Losing weight induces fat cells—remember those?—to produce fewer cytokines. The same goes for regular exercise, 30 minutes a day most days of the week. Flossing combats gum disease, another source of chronic inflammation. Fruits, vegetables, and fish are full of substances that deactivate free radicals.
So if you want to stop inflammation, get off the couch, put your head in the green market, and try not to stub your toe along the way.
If you want to know a 100% natural product to prevent cellular inflammation, we recommend SOUL




